Canvas Equation Editor Tips: Chemistry Using LaTeX
In the Canvas Equation Editor, available in the Rich Content Editor you can optionally use LaTeX code to enter chemical formulas and chemical equations. This document provides tips for entering chemical formula, chemical equations, and scientific notation using LaTeX.
You can learn more about using the Equation Editor.
Notes:
- In addition to the Equation Editor, you can manually enter equations in any Canvas area that includes a text field, such as the title of an assignment or in a calendar event.
- Other resource documents provide tips for entering advanced math.
Writing Chemical Formulas
- For entering a formula, use _ (underscore) for subscripts and ^ (carat) for superscripts. If there is more than one character in the subscript or superscript, wrap the characters with braces { }.
- If you’d like your chemical formulas without the italics, use \text{ } to enclose the elements.
|
Formula |
LaTeX Code |
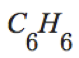
|
C_6H_6 |
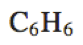
|
\text{C}_6\text{H}_6 |
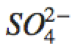
|
SO_4^{2-} |
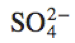
|
\text{SO}_4^{2-} |
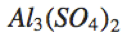
|
Al_3(SO_4)_2 |
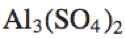
|
\text{Al}_3(\text{SO}_4)_2 |
Writing Scientific Notation
- There are two ways to write the times symbol: \times and \cdot. These are available on the Equation Editor toolbar, but you can also use LaTeX to enter them.
- You can use ^ for exponents. If the exponent contains more than one character, place the exponent text in braces { }.
- To remove italicization, use \text{ } wrapped around units.
- To place space between numbers and units, use \space.
|
Scientific Notation |
LaTeX Code |
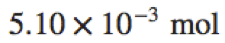
|
5.10\times10^{-3}\space\text{mol} |
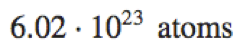
|
6.02\cdot10^{23}\space\text{atoms} |
Writing Chemical Equations
To write chemical equations, you’ll need to use a variety of reaction arrows. Arrows are available on the Equation Editor toolbar, but you can also enter them via LaTeX. Here is a short list of arrows and their LaTeX codes:
|
Arrow |
LaTeX Code |

|
\longrightarrow |

|
\longleftarrow |

|
\xrightarrow{text} |
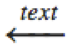
|
\xleftarrow{text} |
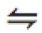
|
\leftrightharpoons |
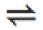
|
\rightleftharpoons |
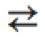
|
\rightleftarrows |
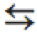
|
\leftrightarrows |
Using LaTeX, you can enter a variety of equations. You may wish to use \space to provide space between quantities and chemical formulas. If you want to remove the italicized elements, it is usually easier to write the formula first, and then go through and insert \text{ } around all the elements in the formula.
|
Scientific Notation |
LaTeX Code |

|
CH_4+2O_2\longrightarrow CO_2+2H_2O |
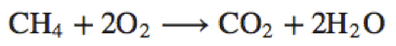
|
\text{CH}_4+2\text{O}_2\longrightarrow \text{CO}_2+2\text{H}_2\text{O} |

|
2 H_2O_2\space(aq)\xrightarrow{Pt} 2 H_2O\space(l)+O_2\space(g) |
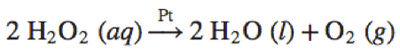
|
2\space\text{H}_2\text{O}_2\space(aq) \xrightarrow{\text{Pt}} 2\space\text{H}_2\text{O}\space(l)+ \text{O}_2\space(g) |
Last update: 2024-04-22
You can access this resource from the following Canvas Guide:
